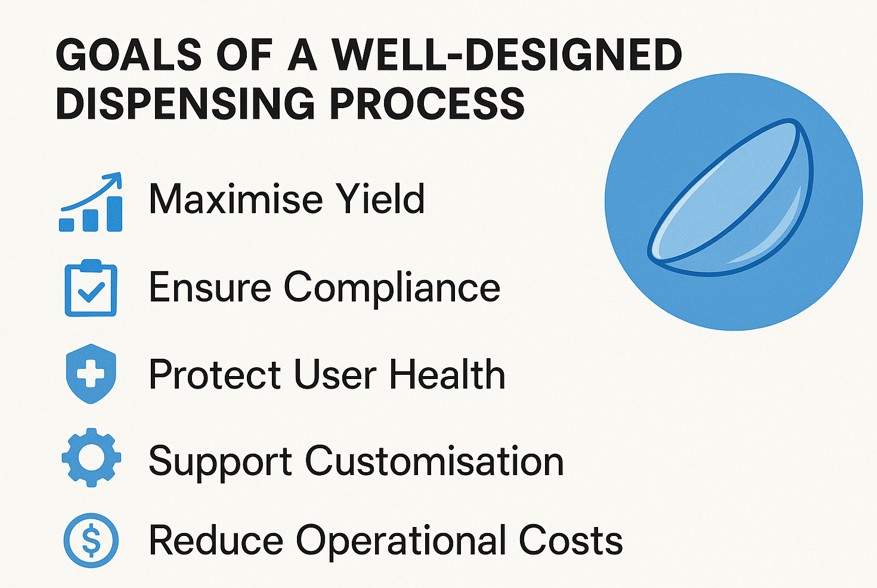
Ever wonder how those tiny lenses that give you crystal-clear vision are made? It’s a fascinating process where precision is key! In contact lens manufacturing, dispensing isn’t just a step—it’s the backbone of quality, safety, and comfort. From the moment raw materials are measured to the final packaging, accurate micro-dispensing is what connects innovative design with reliable performance.
Accurate Material Application
Think of it like baking a cake – the right amount of each ingredient makes all the difference.

- Uniform Lens Formation: Precise dispensing is crucial in determining the lens’s shape, size, and thickness, all of which are key to wearer comfort and clear vision, whether using hydrogel or silicone hydrogel.
- Defect Prevention: Inaccurate dispensing can lead to weaknesses, bubbles, or warping, resulting in product failure.
Ensuring Correct Parameters (Shape, Size & Power)
It’s like getting a tailored suit – it has to fit just right.
- Personalised Lenses: Precise volumetric control ensures that lenses meet prescription-specific parameters, such as curvature, diameter, and power.
- Batch Uniformity: Automation ensures that every lens within a batch performs identically, which is vital for brand integrity and user trust.
Quality Control

- Precision Dosing: Eliminating over- or under-dispensing helps to avoid variability in lens quality.
- Material Uniformity: Accurate control prevents bubbles, stringing, or opacity, all common causes of rejection.
Sterility & Safety
- Sterile Processing: Fluids, such as preservatives and packing solutions, must be dosed in aseptic conditions to prevent microbial contamination.
- Sealed Hydration: Precise volumes of sterile liquid in packaging maintain hydration and prevent blister seal failure during heat sealing.

Driving Cost Efficiency Through Precision

- Reduced Waste: Avoiding material overuse in monomer dispensing saves on raw material costs.
- Minimised Rejects: Better dosing control results in fewer quality rejections and reduced returns.
Comfort & Safety
- Proper Fit: Volumetric accuracy ensures lenses aren’t too thick, thin, or misshapen, preventing corneal irritation or discomfort.
- Hydration & Breathability: Accurate fluid volumes and compositions help maintain water content and oxygen permeability, crucial for eye health.
Regulatory Compliance
- Global Standards: Compliance with FDA 21 CFR, ISO 13485, and EU MDR depends on reproducibility and traceability in production processes.
- Batch Traceability: Automated dispensing systems often include data logging, allowing for traceability in audits or recalls.
Long-Term Lens Performance
- Durability & Optical Clarity:
- Uniformly dispensed monomers and coatings ensure lenses remain clear, flexible, and effective over time.
Fluids Involved in Contact Lens Manufacturing
Here’s a quick look at the liquids that make it all happen:
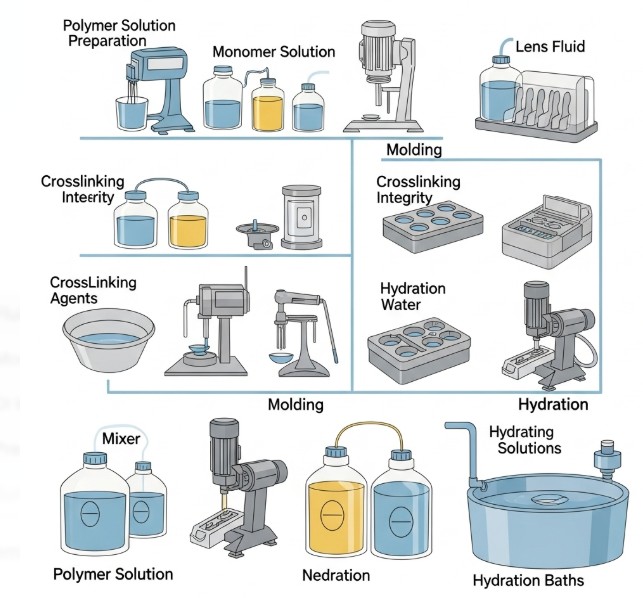
A variety of fluids are dispensed during production, each with its unique challenges:
- Monomers & Crosslinkers: To form the lens matrix (hydrogels).
- DI Water: To hydrate, rinse, or remove unreacted monomer.
- Preservative Solutions: To maintain sterility during storage.
- Surfactants & Solvents: For cleaning or surface treatment processes.
Addressing Micro-dispensing Challenges
- Viscosity & Rheology
- Material Compatibility
- Environmental Control
- Microbubble Elimination

Let’s Talk
Want to improve yield, reduce defects, or achieve regulatory-ready consistency in your contact lens manufacturing process?
📧 Contact info@industrial-fluidics.com